 Boltzmann's entropy equation was: S kBlnt, where: t is the distribution function for the microstates in a system. /abstract-tendril-particles-898633556-5a5795ab7bb28300374e1d9b.jpg)
I think it's best if you stick to just using the value of the standard molar entropy as seen in your textbook appendix, not calculating it from scratch. Boltzmanns entropy equation was: S kBlnt, where: t is the distribution function for the microstates in a system. 9.47 to calculate the change in entropy between the two temperatures. 
by Vic 3 answersTriple point pressure: S gas,1 bar: Entropy of gas at standard. CPo is Thus, the molar if we heat measure capacity the of the system at constant. I think its best if you stick to just using the value of the standard molar entropy as seen in your textbook appendix, not calculating it from scratch. It is also known as methyl fluoride and has a molar mass of 34.03 g/mol. Note: ρ is density, n is refractive index at 589 nm, and η is viscosity, all at 20 ☌ T eq is the equilibrium temperature between two phases: ice/liquid solution for T eq < 0–0.1 ☌ and NaCl/liquid solution for T eq above 0.1 ☌. Well, it's a lot more complicated than it seems. Well, its a lot more complicated than it seems. Thermodynamic properties Phase behaviorĭensity data of aqueous solutions Water–NaCl phase diagram Properties of water–NaCl mixtures NaCl, wt% Structure and properties Structure and properties White Rose Research Online URL for this paper. It is highly recommended that you seek the material safety data sheet ( MSDS) for this chemical from a reliable source such as eChemPortal, and follow its direction. This is a repository copy of Estimation of standard molar entropy of cement hydrates and clinker minerals. The handling of this chemical may incur notable safety precautions. Mass concentration, g/(100 ml) at 15.This page provides supplementary chemical data on sodium chloride. Standard molar entropy, S o liquid: 200.4 J/(mol K) Enthalpy of combustion, c H o 1785.7 kJ/mol Heat capacity, c p: 125.5 J/(mol K) Gas properties Std enthalpy change of formation, f H o gas: 218.5 kJ/mol Standard molar entropy, S o gas: 295.35 J/(mol K) Heat capacity, c p: 75 J/(mol K) van der Waals constants: a 1409.4 L 2 kPa. Properties of aqueous ethanol solutions ĭata obtained from Lange 1967 Mass fraction, Uses formula P mm Hg = 10 8.04494 − 1554.3 222.65 + T ĭensity of ethanol at various temperatures 
Shomate equation parameters for liquid phase thermochemical functions. 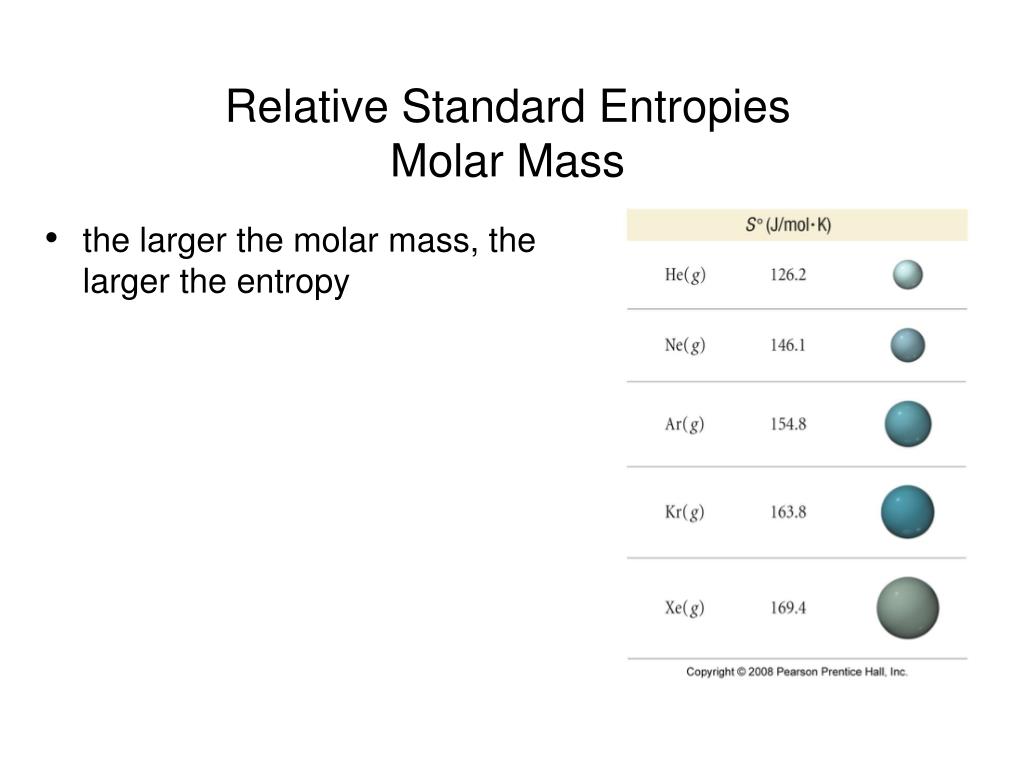
Liquid phase molar entropy at standard conditions, 1 bar or 1 atm standard pressure check source for exact conditions (S° liquid). Vapor pressure of liquid P in mm HgĮthanol vapor pressure vs. Liquid phase molar entropy at standard conditions, 1 bar standard pressure (S° liquid,1 bar). Entropy of gas at standard conditions (1 bar) Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. the entropy of a pure substance at 298 K and 1 atm pressure). Keywords Entropy Carbon monoxide Hydrogen chloride Hydrogen fluoride Deuterium fluoride Boron monobromide Nitrogen monoxide 1. Present calculation scheme is useful to calculate molar entropy for other gaseous substances. Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. Molar entropies of six gaseous substances are excellently predicted. Standard molar entropy The total (minimal) amount of entropy that 1 mole of a substance gains, as it is brought from 0K to the standard conditions is known. This page provides supplementary chemical data on ethanol.Įxternal MSDS Structure and properties Structure and propertiesĠ.1660 W m −1 K −1 (saturated liquid at 300 K) Standard Molar Entropy, S 0 The entropy of a substance has an absolute value of 0 entropy at 0 K. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
